29 Apr Do same-purpose autoinjectors perform the same?
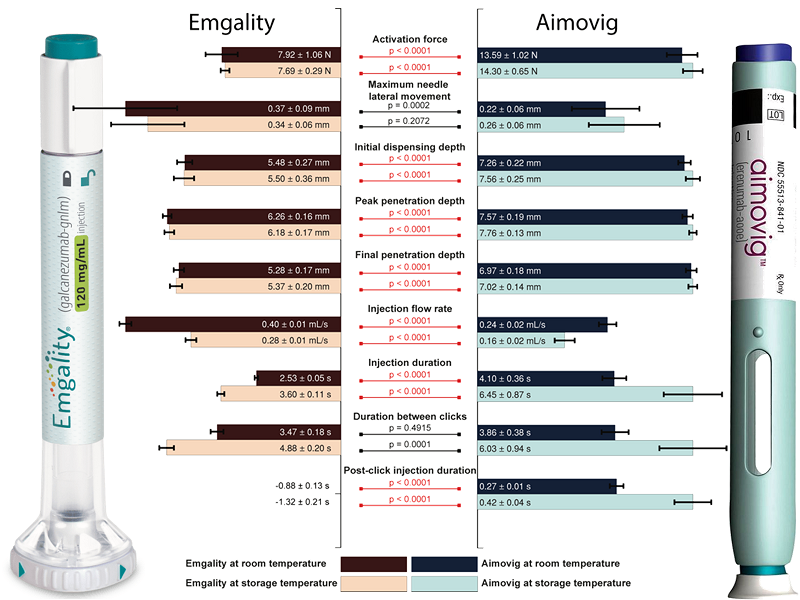
Autoinjectors are drug delivery medical devices for self-administered injection of biopharmaceuticals. Even though the working principle of single-spring-actuated autoinjectors is similar, differences in device design can affect the functionality and performance. From an engineering point of view, this study investigates the performance differences of two single-spring-actuated autoinjectors – Emgality and Aimovig. We found several statically significant kinematic performance differences between the two. Yet to be further verified in clinical trials, these differences may result in different user experiences, risk of intramuscular injection, and premature device removal.

Our measurements indicate that: 1) the Emgality device requires the user to exert a lower force on the button for device activation, 2) the Emgality device shows higher lateral needle movement during insertion, 3) both Emgality and Aimovig devices have injection depth beyond dermis layer with Emgality noticeably shallower, and less likely to enter IM layer, 4) the Emgality device completes injection in a shorter time, and the device-to-device variability is less at both test conditions, 5) the Aimovig device sends out premature dose confirmation “click” sound before actual dose completion, which is undesired and may result in decreased user confidence of dose confirmation. The difference in performance between room temperature and storage temperature tests revealed that only the injection flow rate and duration are significantly affected for both devices. The increase in variability at the storage temperature was observed for the Aimovig device.



No Comments